 Oxygen is also a component of hundreds of thousands of organic compounds.Įstimated Crustal Abundance: 4.61×10 5 milligrams per kilogramĮstimated Oceanic Abundance: 8. Molecular oxygen, or oxygen gas, has the formula O 2 and is therefore diatomic (has two atoms). Ozone (O 3) forms a thin, protective layer around the earth that shields the surface from the sun's ultraviolet radiation. Oxygen is element number 8 on the periodic table of elements. Liquid oxygen, when combined with liquid hydrogen, makes an excellent rocket fuel. Element Oxygen (O) 8 in Group 16 and p-block with information about Its properties, chemical reaction, biological roles, health effects, sources, history. Oxygen can also be combined with acetylene (C 2H 2) to produce an extremely hot flame used for welding. Carbon is the cosmic product of the burning of helium, in which three helium nuclei, atomic weight 4, fuse to produce a carbon nucleus, atomic weight 12. Oxygen makes up about one fifth of the atmosphere. Scientists use symbols to stand for the chemical elements. It is one of the main elements that make up air, and it is necessary for the survival of all plants and animals. Impurities in molten pig iron are burned away with streams of high pressure oxygen to produce steel. Only hydrogen, helium, oxygen, neon, and nitrogen are atomically more abundant in the cosmos than carbon. Introduction Oxygen is the most common chemical element found on or in Earth. 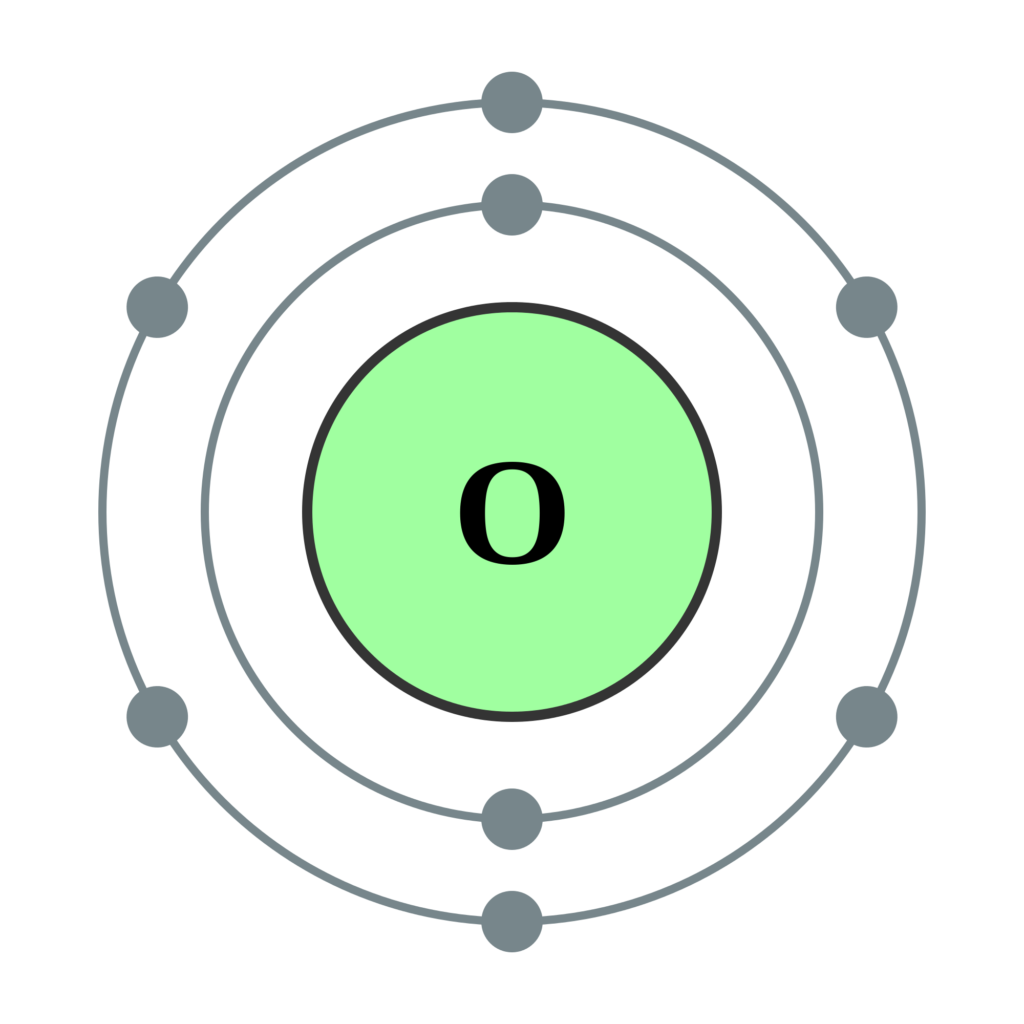
Click the tabs at the top to explore each section. It is required by most living organisms and for most forms of combustion. 1234567LanthanidesActinides Solid Liquid Gas Unknown 4000Kelvin 4000Celsius Periodic Table The Royal Society of Chemistry's interactive periodic table features history, alchemy, podcasts, videos, and data trends across the periodic table. Oxygen is a highly reactive element and is capable of combining with most other elements. Oxygen can also be produced through the electrolysis of water or by heating potassium chlorate (KClO 3). Large amounts of oxygen can be extracted from liquefied air through a process known as fractional distillation.  
Oxygen accounts for nearly half of the mass of the earth's crust, two thirds of the mass of the human body and nine tenths of the mass of water. Oxygen is the third most abundant element in the universe and makes up nearly 21% of the earth's atmosphere. The name oxygen was created by Antoine Lavoisier who incorrectly believed that oxygen was necessary to form all acids. Priestley called the gas produced in his experiments 'dephlogisticated air' and Scheele called his 'fire air'. They were both able to produce oxygen by heating mercuric oxide (HgO). Joseph Priestley and Carl Wilhelm Scheele both independently discovered oxygen, but Priestly is usually given credit for the discovery. Oxygen had been produced by several chemists prior to its discovery in 1774, but they failed to recognize it as a distinct element. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
